- Research Article
- Open access
- Published:
Pushing it to the Limit: Adaptation with Dynamically Switching Gain Control
EURASIP Journal on Advances in Signal Processing volume 2007, Article number: 051684 (2006)
Abstract
With this paper we propose a model to simulate the functional aspects of light adaptation in retinal photoreceptors. Our model, however, does not link specific stages to the detailed molecular processes which are thought to mediate adaptation in real photoreceptors. We rather model the photoreceptor as a self-adjusting integration device, which adds up properly amplified luminance signals. The integration process and the amplification obey a switching behavior that acts to shut down locally the integration process in dependence on the internal state of the receptor. The mathematical structure of our model is quite simple, and its computational complexity is quite low. We present results of computer simulations which demonstrate that our model adapts properly to at least four orders of input magnitude.
References
Walraven J, Enroth-Cugell C, Hood D, MacLeod D, Schnapf J: The control of visual sensitivity: receptoral and postreceptoral processes. In The Neurophysiological Foundations of Visual Perception. Edited by: Spillman L, Werner J. Academic Press, New York, NY, USA; 1990:53–101.
Barlow H, Levick W: Threshold setting by the surround of cat retinal ganglion cells. Journal of Physiology, London, B 1976, 212: 1.
Hood D, Finkelstein M: Sensitivity to light. In Handbook of Perception and Visual Performance, Volume 1: Sensory Processes and Perception. Edited by: Boff K, Kaufman L, Thomas J. John Wiley & Sons, New York, NY, USA; 1986:5.1–5.66. chapter 5
Mante V, Frazor RA, Bonin V, Geisler WS, Carandini M: Independence of luminance and contrast in natural scenes and in the early visual system. Nature Neuroscience 2005,8(12):1690–1697. 10.1038/nn1556
van Hateren JH: Processing of natural time series of intensities by the visual system of the blowfly. Vision Research 1997,37(23):3407–3416. 10.1016/S0042-6989(97)00105-3
Martin G: Schematic eye models in vertebrates. Progress in Sensory Physiology 1983, 4: 44.
Shapley R, Enroth-Cugell C: Visual adaptation and retinal gain controls. Progress in Retinal Research 1984, 3: 263–346.
Laughlin SB: The role of sensory adaptation in the retina. Journal of Experimental Biology 1989, 146: 39–62.
Normann R, Perlman I, Hallet P: Cone photoreceptor physiology and cone contributions to colour vision. In Vision and Visual Dysfunction, The Perception of Colour. Edited by: Gouras P. Macmillan Press, London, UK; 1991:146–162.
Barlow HB: The Ferrier Lecture, 1980. Critical limiting factors in the design of the eye and visual cortex. Proceedings of the Royal Society of London. Series B. Biological Sciences 1981,212(1186):1–34. 10.1098/rspb.1981.0022
Hood DC: Lower-level visual processing and models of light adaptation. Annual Review of Psychology 1998, 49: 503–535. 10.1146/annurev.psych.49.1.503
Meister M, Berry MJ II: The neural code of the retina. Neuron 1999,22(3):435–450. 10.1016/S0896-6273(00)80700-X
Fahrenfort I, Habets RL, Spekreijse H, Kamermans M: Intrinsic cone adaptation modulates feedback efficiency from horizontal cells to cones. Journal of General Physiology 1999,114(4):511–524. 10.1085/jgp.114.4.511
Fain GL, Matthews HR, Cornwall MC, Koutalos Y: Adaptation in vertebrate photoreceptors. Physiological Reviews 2001,81(1):117–151.
Burkhardt DA: Light adaptation and photopigment bleaching in cone photoreceptors in situ in the retina of the turtle. Journal of Neuroscience 1994,14(3 I):1091–1105.
Dowling J: The Retina: An Approachable Part of the Brain. Belknap Press/Havard University Press, Cambridge, Mass, USA; 1987.
Kolb H, Fernandez E, Nelson R: Webvision. The organization of the vertebrate retina. 2000.https://doi.org/retina.umh.es/Webvision
Burns ME, Baylor DA: Activation, deactivation, and adaptation in vertebrate photoreceptor cells. Annual Review of Neuroscience 2001, 24: 779–805. 10.1146/annurev.neuro.24.1.779
Grossberg S, Hong S: Cortical dynamics of surface lightness anchoring, filling-in, and perception. Journal of Vision 2003,3(9):415a.
Hong S, Grossberg S: A neuromorphic model for achromatic and chromatic surface representation of natural images. Neural Networks 2004,17(5-6):787–808. 10.1016/j.neunet.2004.02.007
Carpenter GA, Grossberg S: Adaptation and transmitter gating in vertebrate photoreceptors. Journal of Theoretical Neurobiology 1981, 1: 1–42.
Kamermans M, Fahrenfort I, Schultz K, Janssen-Bienhold U, Sjoerdsma T, Weiler R: Hemichannel-mediated inhibition in the outer retina. Science 2001,292(5519):1178–1180. 10.1126/science.1060101
Lamb TD: Spatial properties of horizontal cell responses in the turtle retina. Journal of Physiology 1976,263(2):239–255.
Piccolino M, Neyton J, Gerschenfeld HM:Decrease of gap junction permeability induced by dopamine and cyclic adenosine
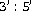 -monophosphate in horizontal cells of turtle retina. Journal of Neuroscience 1984,4(10):2477–2488.
-monophosphate in horizontal cells of turtle retina. Journal of Neuroscience 1984,4(10):2477–2488.Perona P, Malik J: Scale-space and edge detection using anisotropic diffusion. IEEE Transactions on Pattern Analysis and Machine Intelligence 1990,12(7):629–639. 10.1109/34.56205
Calvert PD, Govardovskii VI, Arshavsky VY, Makino CL: Two temporal phases of light adaptation in retinal rods. Journal of General Physiology 2002,119(2):129–145. 10.1085/jgp.119.2.129
Perry RJ, McNaughton PA: Response properties of cones from the retina of the tiger salamander. Journal of Physiology 1991,433(1):561–587.
Calvert PD, Ho TW, Lefebvre YM, Arshavsky VY: Onset of feedback reactions underlying vertebrate rod photoreceptor light adaptation. Journal of General Physiology 1998,111(1):39–51. 10.1085/jgp.111.1.39
Rebrik TI, Korenbrot JI:In intact mammalian photoreceptors,
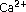 -dependent modulation of cGMP-gated ion channels is detectable in cones but not in rods. Journal of General Physiology 2004,123(1):63–75.
-dependent modulation of cGMP-gated ion channels is detectable in cones but not in rods. Journal of General Physiology 2004,123(1):63–75.Vaquero CF, Pignatelli A, Partida GJ, Ishida AT: A dopamine- and protein kinase A-dependent mechanism for network adaptation in retinal ganglion cells. Journal of Neuroscience 2001,21(21):8624–8635.
Weiler R, Schultz K, Pottek M, Tieding S, Janssen-Bienhold U: Retinoic acid has light-adaptive effects on horizontal cells in the retina. Proceedings of the National Academy of Sciences of the United States of America 1998,95(12):7139–7144. 10.1073/pnas.95.12.7139
Green D, Dowling J, Siegal I, Ripps H: Retinal mechanisms of visual adaptation in the skate. The Journal of General Physiology 1975 ,65(4):483–502. 10.1085/jgp.65.4.483
Barlow HB, Levick WR: Changes in the maintained discharge with adaptation level in the cat retina. Journal of Physiology 1969,202(3):699–718.
Smirnakis SM, Berry MJ, Warland DK, Bialek W, Meister M: Adaptation of retinal processing to image contrast and spatial scale. Nature 1997,386(6620):69–73. 10.1038/386069a0
Hosoya T, Baccus SA, Meister M: Dynamic predictive coding by the retina. Nature 2005,436(7047):71–77. 10.1038/nature03689
Kawamura S: Rhodopsin phosphorylation as a mechanism of cyclic GMP phosphodiesterase regulation by S-modulin. Nature 1993,362(6423):855–857. 10.1038/362855a0
Chen C-K, Inglese J, Lefkowitz RJ, Hurley JB:
 -dependent interaction of recoverin with rhodopsin kinase. Journal of Biological Chemistry 1995,270(30):18060–18066. 10.1074/jbc.270.30.18060
-dependent interaction of recoverin with rhodopsin kinase. Journal of Biological Chemistry 1995,270(30):18060–18066. 10.1074/jbc.270.30.18060Klenchin VA, Calvert PD, Bownds MD: Inhibition of rhodopsin kinase by recoverin. Further evidence for a negative feedback system in phototransduction. Journal of Biological Chemistry 1995,270(27):16147–16152. 10.1074/jbc.270.27.16147
Gross R, Brajovic V: An image preprocessing algorithm for illumination invariant face recognition. In Audio-and Video-Based Biometrie Person Authentication (AVBPA '03), June 2003, Guildford, UK, Springer Lecture Notes in Computer Sciences Edited by: Kittler J, Nixon M. 2688: 10–18.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 2.0 International License ( https://creativecommons.org/licenses/by/2.0 ), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
About this article
Cite this article
Keil, M.S., Vitrià, J. Pushing it to the Limit: Adaptation with Dynamically Switching Gain Control. EURASIP J. Adv. Signal Process. 2007, 051684 (2006). https://doi.org/10.1155/2007/51684
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1155/2007/51684
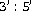 -monophosphate in horizontal cells of turtle retina. Journal of Neuroscience 1984,4(10):2477–2488.
-monophosphate in horizontal cells of turtle retina. Journal of Neuroscience 1984,4(10):2477–2488.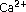 -dependent modulation of cGMP-gated ion channels is detectable in cones but not in rods. Journal of General Physiology 2004,123(1):63–75.
-dependent modulation of cGMP-gated ion channels is detectable in cones but not in rods. Journal of General Physiology 2004,123(1):63–75. -dependent interaction of recoverin with rhodopsin kinase. Journal of Biological Chemistry 1995,270(30):18060–18066. 10.1074/jbc.270.30.18060
-dependent interaction of recoverin with rhodopsin kinase. Journal of Biological Chemistry 1995,270(30):18060–18066. 10.1074/jbc.270.30.18060